Title: The E3 ubiquitin ligase MIEL1 promotes thermo-responsive growth by blocking auto-ubiquitination and self-degradation of XBAT31
Lin-Lin Zhang, Qiang-Hua Zhou, Zhi-Ping Deng, Juan Gao, Hui-Dan Luo, Ying-Ying Tian, Jian-Xiang Liu
Abstract
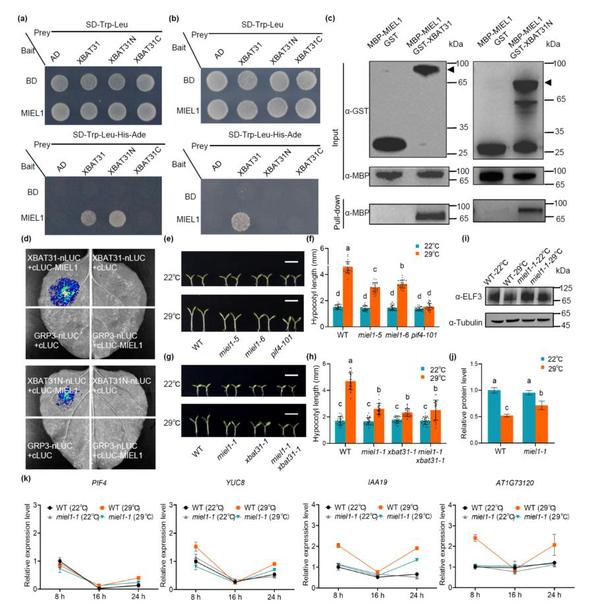
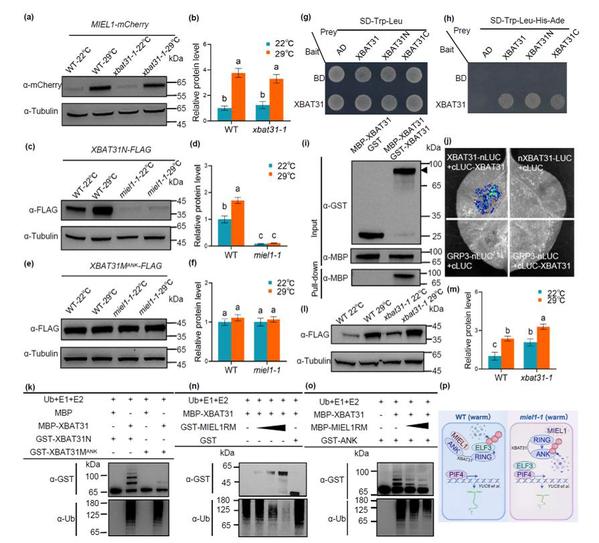
Previous research has revealed that the E3 ubiquitin ligase XBAT31 mediates the ubiquitination and degradation of the thermosensor ELF3, positively regulating hypocotyl elongation under moderately high temperatures. In this study, another E3 ubiquitin ligase, MIEL1, was identified through yeast two-hybrid screening. The findings confirmed that MIEL1 interacts with XBAT31 and the N-terminal region of XBAT31 both in vitro and in vivo. Phenotypic analysis revealed that MIEL1 acts as a positive regulator in thermomorphogenesis, and both MIEL1 and XBAT31 function upstream of the ELF3 signaling pathway. By modulating ELF3 protein levels, they activate PIF4 transcriptional activity, thereby promoting hypocotyl elongation.
Link: https://www.sciencedirect.com/science/article/pii/S2095927326001593