Title: A fibroblast-like endothelial cell state promotes atherosclerosis via CEBPβ-activated TGF-β signaling
Linge Fan, Yingyi Zhu, Yi Li, Zixin Ji, Kefan Ma, Ying Zhang, Leiting Wei, Junbo Chen, Yuanqing Jiang, Dongwu Lai, Lingfeng Qin, Guosheng Fu, Michael Simons, Liang Xu, Luyang Yu , Cong Qiu
Abstract
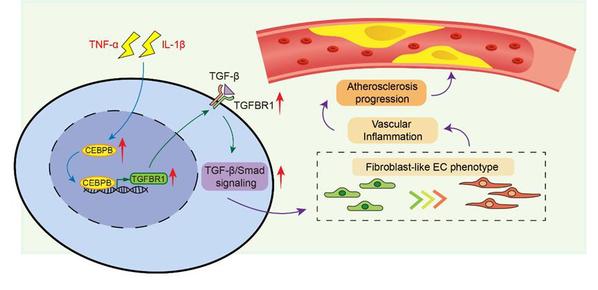
Endothelial cell (EC) dysfunction is a critical driver of chronic vascular inflammation and atherosclerosis. However, the molecular details of EC state dynamics during vascular disease progression remain ill-defined. Here, we used in-depth single-cell RNA sequencing to map transcriptional landscapes and molecular signatures of EC phenotypic plasticity during atherosclerosis in the mouse arota. This analysis identified a unique fibroblast-like EC population in atherosclerotic blood vessels, characterized by high expression of endothelial activation markers and extracellular matrix (ECM) remodeling, which increased with disease severity. Pseudotime trajectory analysis revealed that these fibroblast-like ECs represent terminal states of endothelial-mesenchymal transition (EndMT) during atherosclerosis. Further, the transcription factor C/EBPβ was identified as prominent driver of this phenotype transition as evidenced in vivo and in vitro. Mechanistically, inflammatory cytokines induce C/EBPβ, triggering TGF-β signaling and subsequent regulation of downstream genes via upregulation of TGF-β receptor type I (TGFBR1) through direct interaction with its promoter. Endothelial overexpression of C/EBPβ in vivo exacerbated atherosclerotic plaques, increased vascular inflammation, and elevated endothelial TGFBR1 levels. These findings highlight endothelial C/EBPβ as a novel regulator of TGF-β signaling and pathological fibroblast-like EC phenotypes during atherosclerosis, linking cytokine-driven inflammation with TGF-β-mediated endothelial dysfunction.
Link:https://link.springer.com/article/10.1038/s44318-025-00684-x