Title: Setd4-expressing cells drive regenerative recovery in chronic liver injury
Xi-Zheng Jia, Tian-Tian Zhuang, Ri-Sheng Yang, Yun-Wen Gu, Yi-Zhe Zeng, Christopher Wood, Jin-Shu Yang, Wei-Jun Yang
Abstract
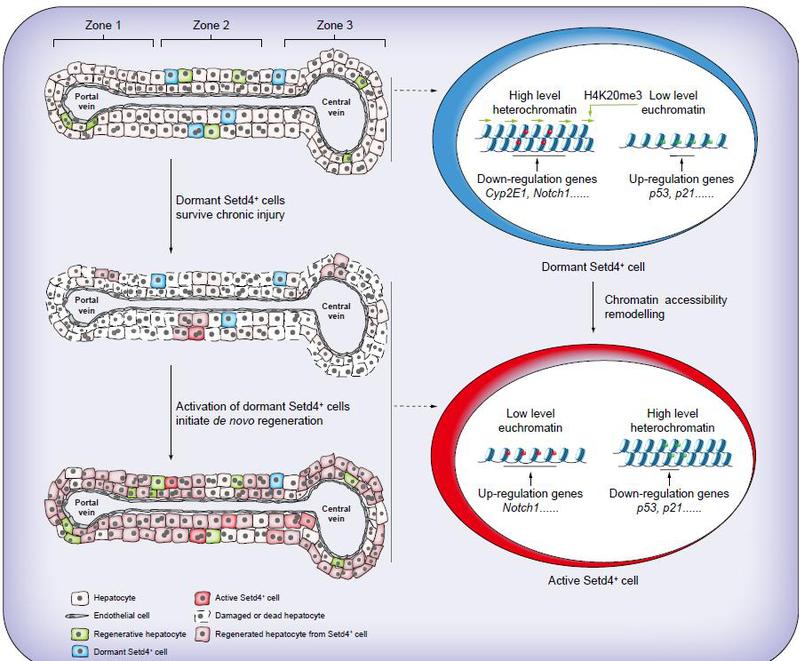
As the metabolic center of the body, the liver is susceptible to chronic damage from toxic metabolites or exogenous substances, leading to the apoptosis of hepatocytes, which subsequently influences the progression of the liver toward end-stage liver diseases such as fibrosis, cirrhosis, or hepatocellular carcinoma. Following chronic liver injury, to preserve liver function, the liver initiates a regenerative process to replenish lost hepatocytes and achieve regenerative repair of chronic liver injury. Even when recovery from chronic injury is achievable, the process relies on de novo cellular regeneration, the core underlying mechanism of which remains to be understood and elucidated. Recently, the laboratory of Yang Weijun at the College of Life Sciences, Zhejiang University, published a research paper titled “Setd4-expressing cells drive regenerative recovery in chronic liver injury” in the Journal of Hepatology. This study reveals a core mechanism underlying chronic liver injury repair: a population of quiescent cells expressing Setd4 resides in zone 2 of the hepatic lobule. These cells maintain a dormant state through H4K20me3-mediated regulation of chromatin architecture and silencing of cellular processes such as proliferation and metabolism, thereby withstanding continuous attack from chronic injury. When injury leads to the exhaustion of proliferative capacity in hepatocytes, these quiescent hepatocytes are triggered to be awakened, activated, proliferated, and differentiated through chromatin accessibility remodeling, replenishing lost hepatocytes and driving regenerative repair of liver tissue. The study proposes a dual-track model of liver injury regeneration: proliferative hepatocytes are responsible for acute liver injury repair, whereas activated quiescent cells mediate regenerative repair of chronic liver injury. By modulating chromatin remodeling in quiescent hepatocytes, endogenous liver damage regeneration can be activated. These findings not only elucidate the cellular and molecular mechanisms of chronic liver injury repair but also lay a theoretical foundation for the development of innovative therapies aimed at awakening quiescent cells, potentially reshaping the clinical treatment landscape for end-stage liver diseases in the future.
Link: https://doi.org/10.1016/j.jhep.2025.09.034